Ammonia Electrical Conductivity
Please contact the SmartMeasurement factory for assistance. Solar Radiation.
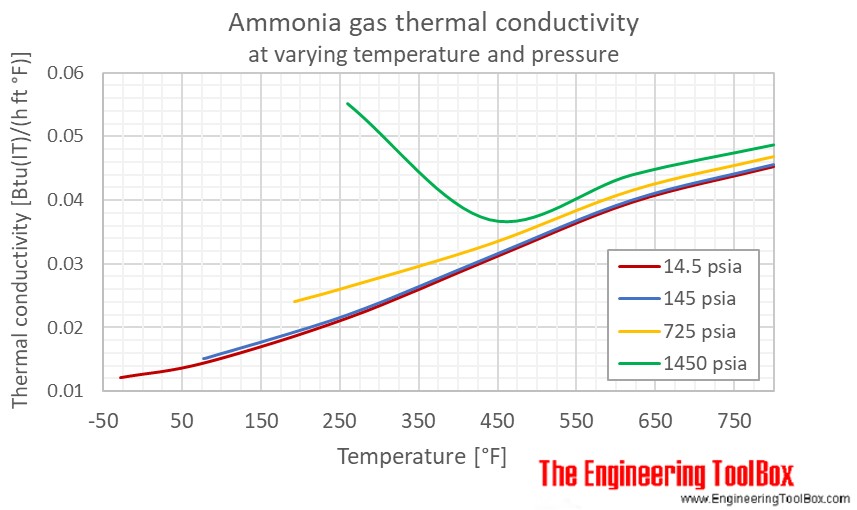
Ammonia Thermal Conductivity Vs Temperature And Pressure
Conductivity values displayed in yellow are questionable.

. However as the water becomes more alkaline the pH increases ammonia becomes progressively more poisonous to these same organisms. Temperature and Pressure - Online calculator figures and tables showing thermal conductivity of liquid and gaseous ammonia at temperatures ranging -70 to 425 C -100 to 800 F at atmospheric and higher pressure - Imperial and SI Units. Water temperature can play a role in the shift between ammonium and ammonia in water.
These cells were tested with ammonia and hydrogen and their results showed good performance with ammonia with a power density of 3150 Wm 2 compared to 3350 Wm 2 produced when using H 2 and both producing voltages of 095 V. Phosphorus and Nitrogen as Nitrate and Ammonia. Pure water containing no exogenous ions is an excellent electronic insulator but.
Ammonia - Thermal Conductivity vs. Electrical infrastructure assets including reticulation assets substations switch gear and transformers 25 years. Turbidity Total Suspended Solids and Clarity.
Ammonia - Thermal Conductivity vs. Although an Ag layer was used to improve the. PH is the negative log.
Measurement methods and technology can be found in the Measurements section. Offices at 1-866-404-5415 for assistance. Similar work has been carried out by other institutions using other ceramic composites and ammonia blends.
K 2 SO 4 990 was added to ensure sufficient ionic conductivity keeping the total concentration of K to be 01 M and provides SO 4 2 ions as an external Raman reference. In addition to its effects on aquatic organisms high water temperatures can increase the solubility and thus toxicity of certain compounds 1These elements include heavy metals such as cadmium zinc and lead as well as compounds like ammonia 1920Water temperature can not only. Conductivity Salinity and Total Dissolved Solids.
Generally larger ions move slower than smaller ions and so contribute less to the conductivity. However they do not show anomalous thermodynamic kinetic or structural properties like those observed in water because none of them can form four hydrogen bonds. However the performance of ammonia synthesis by this reactor prototype was far from satisfactory in which ammonia generation was observed from 500 C and the rate of production was increased by two orders of magnitude at 650 C.
The electrical conductivity EC of water is a measure of the ability of a solution to carry or conduct an electrical current. Ammonia is a compound of nitrogen and hydrogen with the formula NH 3A stable binary hydride and the simplest pnictogen hydride ammonia is a colourless gas with a distinct pungent smell. Biologically it is a common nitrogenous waste particularly among aquatic organisms and it contributes significantly to the nutritional needs of terrestrial organisms by serving as a.
Temperature and Pressure - Online calculator figures and tables showing thermal conductivity of liquid and gaseous ammonia at temperatures ranging -70 to 425 C -100 to 800 F at atmospheric and higher pressure - Imperial and SI Units. Other molecules like hydrogen fluoride ammonia and methanol can also form hydrogen bonds. They attributed this to Rus very poor electrical conductivity.
Electrical conductivity is dependent on all the ions that are in solution. For instance ammonia is relatively harmless to fish in neutral or acidic water. If a particular chemical is not listed or if you would like application assistance with an electromagnetic flowmeter application please feel free to contact SmartMeasurement TM s US.
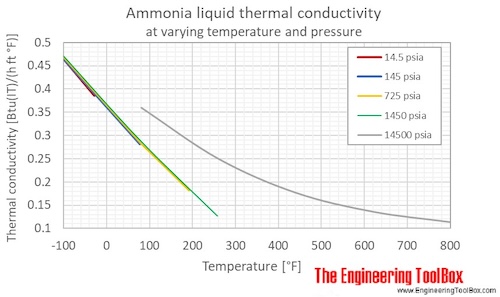
Ammonia Thermal Conductivity Vs Temperature And Pressure
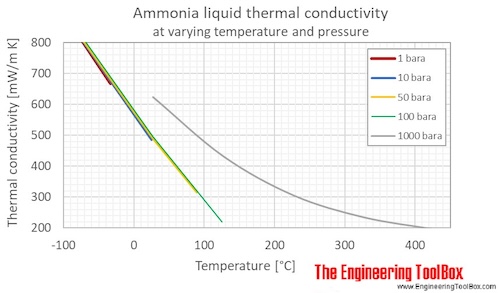
Ammonia Thermal Conductivity Vs Temperature And Pressure

Physical Properties Ammonia 7 Physical Properties Physics Ammonia

Electrical Conductivity And Density Of Ammonium Nitrate Formamide Mixtures Journal Of Chemical Engineering Data
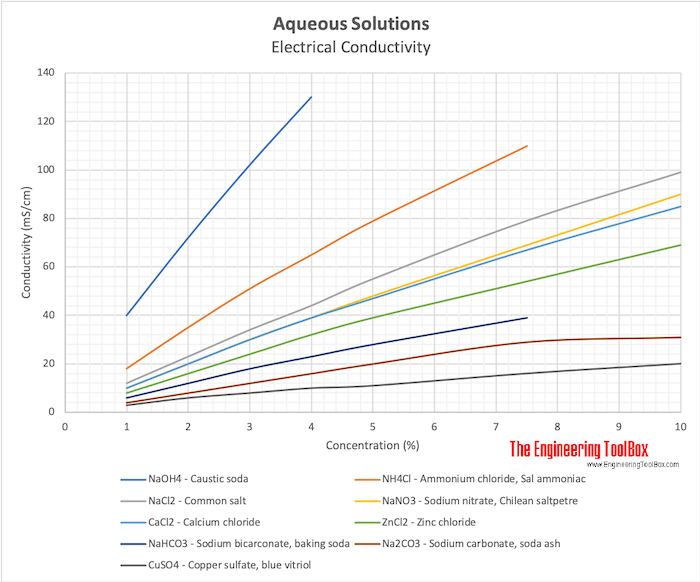
Electrical Conductivity Elements And Other Materials

Properties Ionic Covalent Compounds Experiment 2 Electrolytes Ionic Chemical Changes
Comments
Post a Comment